Tumor Mutational Burden for Immunotherapy Response
Tumor mutational burden (TMB) is gaining tremendous interest in recent years as a biomarker particularly for predicting response to immuno-oncology (IO) therapies. TMB is measured as the total number of mutations per megabase within the coding region of a tumor genome. Tumors with high TMB are likely to harbor more proteins with mutations, which are presented as neoantigens that can be recognized by T cells. Patients with high TMB have been found to have more favorable response to checkpoint inhibitors, such as atezolizumab and pembrolizumab, than those with lower TMB.
The gold standard for measuring TMB has been whole exome sequencing (WES). Recent studies have demonstrated that targeted NGS panels can be an effective and economical method for estimating TMB, provided that the panels contain at least 300 genes and cover more than 1 Mb of genomic content. Amplicon-based targeted NGS panels require smaller DNA input amounts and provide a simpler and faster workflow, allowing sequencing to be carried out on the same day for rapid measurement of TMB. However, researchers should be mindful when selecting the appropriate technology for assay development since not all amplicon-based target enrichment technologies can interrogate hundreds of genes in a single assay.
CleanPlex® TMB Panel
Rapid Measurement of Tumor Mutational Burden
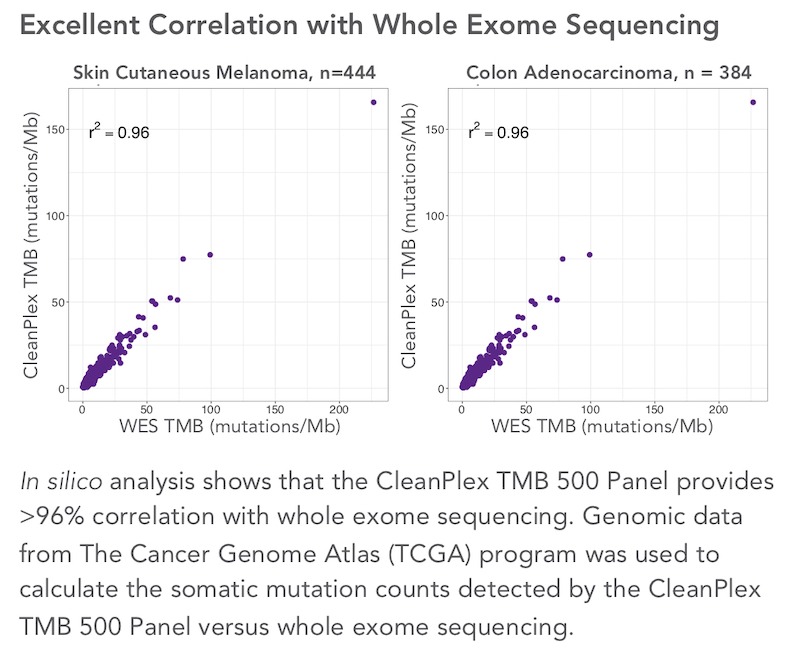
Paragon Genomics has developed a high-performance CleanPlex TMB 500 Gene Panel that contains over 500 genes covered by approximately 27,000 amplicons. One custom version of the panel was tested by Berry Oncology (affiliated with Berry Genomics) to measure TMB in solid tumor FFPE samples. For each sample, the TMB was calculated, the types of mutations were documented, and the top mutated genes were identified. They compared the mutations identified using the TMB panel to those identified with WES for the same 103 samples and found a high degree of overlap between the two methods. They also found a high degree of correlation when they compared the TMB calculated using the two methods.
High TMB Correlation with Whole Exome Sequencing Data
Poster
Assessment of Tumor Mutational Burden using a Rapid, Amplicon-Based NGS Target Enrichment Strategy
Download our poster from the 2019 AMP Annual Meeting to see data demonstrating CleanPlex technology’s ability to rapidly measure tumor mutational burden.
Request a Quote
Request Customization
If the content of the CleanPlex TMB 500 Panel isn’t exactly what you need, we can also design a custom TMB panel for you. CleanPlex® Custom NGS Panels are powered by Paragon Genomics’ CleanPlex Technology – an ultra-high multiplex PCR-based target enrichment technology for next-generation sequencing (NGS). It features a highly advanced proprietary primer design algorithm and an innovative, patented background cleaning chemistry. Together they enable more than 20,000 amplicons to be multiplexed in a single assay to build NGS panels as large as a few megabases in size for interrogating hundreds of genes.
At Paragon Genomics, we take your privacy and data very seriously. You can review our Privacy Policy online or download the PDF.