Respiratory Viral Kit: CleanPlex® Respiratory Virus Research Kit Combining Influenza A/B, RSV A/B, and SARS-CoV-2 Testing
Paragon Genomics released CleanPlex SARS-CoV-2 Research and Surveillance Kit in early 2020 for ultra-sensitive detection and complete genomic sequencing of the SARS-CoV-2 virus responsible for the COVID-19 pandemic. Throughout flu season, there is a need for assaying common respiratory viruses concurrently. The CleanPlex Respiratory Virus Research Kit combines the original SARS-CoV-2 whole-genome sequencing with expertly designed primers specific to influenza A subtypes H1N1, H1N2, H3N2, influenza B, and respiratory syncytial virus (RSV) A&B.
Product Highlight
CleanPlex Respiratory Virus Research Kit combines the original SARS-CoV-2 whole-genome sequencing with expertly designed primers specific to influenza A subtypes H1N1, H1N2, H3N2, influenza B, and respiratory syncytial virus (RSV) A&B.
View ProductCleanPlex Respiratory Virus Research Panel: What is a Respiratory Virus Panel?
Our amplicon-based respiratory viral panel covers the entire SARS-CoV-2 viral genome and 149 strategically selected characteristic regions of a variety of viral segments for highly confident detection and identification. It is designed for COVID-19, influenza (flu A/B), and RSV (A/B) detection, research, and surveillance. This panel enables complete genome sequencing of the SARS-CoV-2 virus and concurrent influenza and RSV sub-typing with highly sensitive detection.
The NGS panel not only allows detection and differentiation of the respiratory tract viruses but also enables subtyping and potential strain information for mutation analysis and informed infection control through sequence information.
Compared to most qPCR-based methods, this NGS-based panel covers many more targets for higher sensitivity and is suitable for higher throughput sample pooling on Illumina and MGI sequencers related to the respiratory syncytial virus, influenza A subtypes H1N1, H1N2, H3N2, and influenza B.
The respiratory virus primers were expertly designed in consideration of over 45,000 viral genome sequences to generate highly specific, but also universally conserved targets for each influenza and RSV subtype to allow confident detection throughout flu seasons, regardless of changes in strain prevalence.
Built upon the CleanPlex SARS-CoV-2 Research and Surveillance Panel, the Respiratory Virus Research Panel v2 addresses the need to assay the SARS-CoV-2 and other respiratory viruses concurrently during the overlapping flu season and the COVID-19 pandemic.
Dr. Xiaowu Gai, Director of Bioinformatics at the Center for Personalized Medicine in the Department of Pathology and Laboratory Medicine at Children’s Hospital Los Angeles, and Associate Professor of Clinical Pathology at the Keck School of Medicine at the University of Southern California, said, “I was very impressed by how well the CleanPlex SARS-CoV-2 panel worked when we evaluated it back in March. Working with Paragon Genomics, we were able to leverage the panel to quickly launch a SARS-CoV-2 whole-genome sequencing assay and used it effectively for a number of genomic epidemiology studies.”
The CleanPlex® Respiratory Virus Research Kit utilizes the CleanPlex multiplex PCR-based targeted sequencing technology to provide an easy-to-use, fast, and comprehensive solution for detection and differentiation of these common viruses for informed infection control through comprehensive sequence information.
Highlights of the CleanPlex Respiratory Virus Research Panel
Significant Cost Savings: 99% reduction in sequencing cost. Only 30K–50K reads per sample required v.s. 10M reads required by many metagenomics sequencing methods.
High Coverage of Target Regions: Whole-genome coverageof the SARS-CoV-2 virus and selective coverage of the influenza virus and RSV gene segments for sub-typing.
Ultra-Sensitive Detection: Multiplex PCR based method for more sensitive detection of low viral copies.
Fast, Streamlined Workflow: Generate sequencing-ready libraries in just 6 hours using a rapid, four-step protocol including RT step with minimal hands-on time.
Superb Performance: Prepare high-quality NGS libraries with excellent on-target performance using CleanPlex Technology to enable efficient use of sequencing reads. CleanPlex panels’ have higher on target and sensitivity compared to similar hybrid captured-based target enrichment panels.
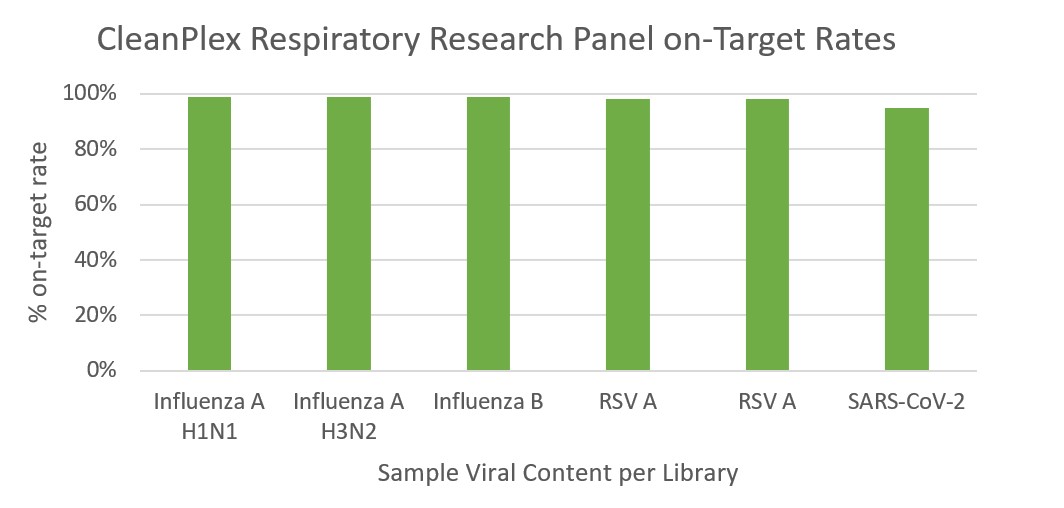
CleanPlex Respiratory Virus Research Panel Performance
CleanPlex amplicon-based target enrichment method allows highly specific amplification, resulting in high on-target rates and reduced overall sequencing depth per sample. The specificity table below shows the percentage of amplicons with >30X coverage in libraries prepared from each virus control sample using only 50,000 down-sampled reads per sample. The on-target rates for each sample are also shown in the histogram below. The data demonstrate high coverage and on-target rates for detection even at low sequencing depths.
Product Highlight
The CleanPlex Respiratory Virus Research Kit is designed for the detection, research, and surveillance of COVID-19 SARS-CoV-2 virus, Influenza (flu A/B), and respiratory syncytial virus (RSV) (A/B), enabling complete genome sequencing of the SARS-CoV-2 virus and concurrent influenza and RSV sub-typing with highly sensitive detection.
Download Product Sheet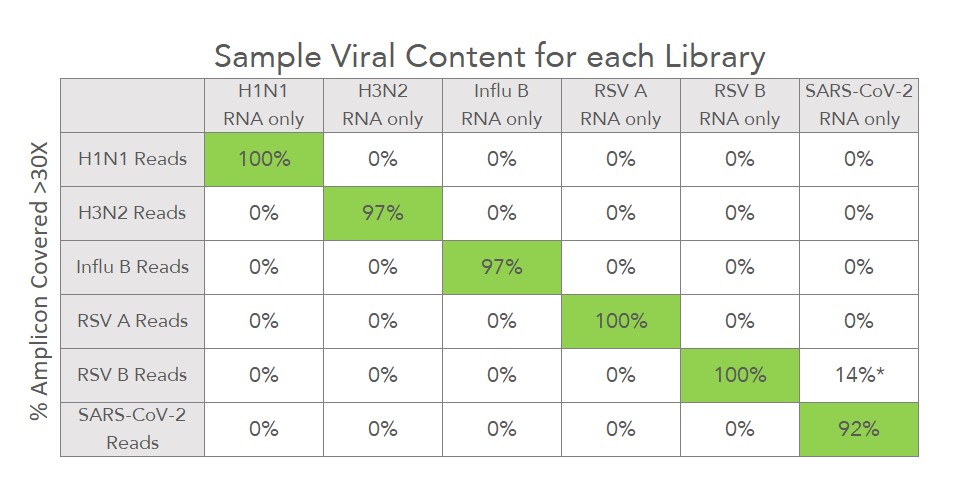
Ordering Your Respiratory Virus Panel Test
The CleanPlex Respiratory Virus Research Panel v2 for respiratory virus sequencing is available today. CleanPlex Indexed PCR Primers and CleanMag® Magnetic Beads are ordered separately to complete the workflow from input RNA to sequencing-ready NGS libraries.
For additional information, visit the product page here.
On-Demand Webinar Videos
Paragon Genomics | Sophia Genetics COVID-19 Webinar
Paragon Genomics | Children’s Hospital Los Angeles COVID-19 Webinar
CONTACT US