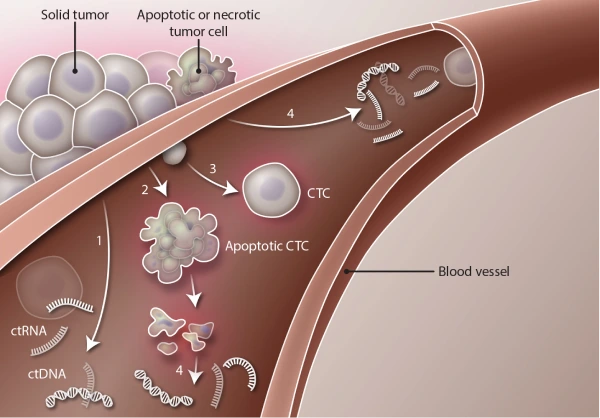
Both circulating tumor DNA (ctDNA) and Circulating Tumor Cells (CTCs) serve as biomarkers for early cancer detection and provide guidance in cancer treatment. However, there are significant differences between them, the most profound differences being the type and stage of tumors from which they are released.
ctDNA is fragments of DNA from apoptotic tumor-derived cells shed into the bloodstream. ctDNA might be useful in the detection, tracking, and treatment of tumors, as its presence can indicate cancer.
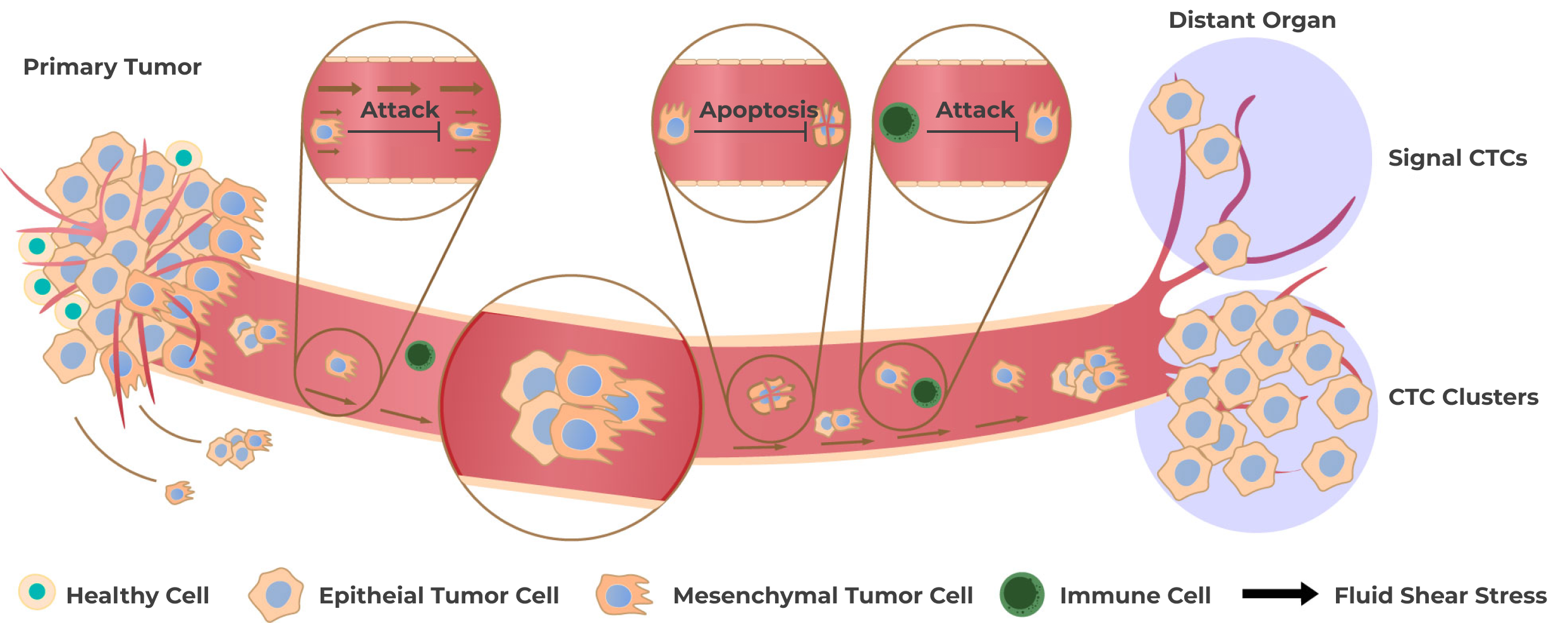
CTCs are released into the bloodstream from primary cancer, metastasis, as well as from a disseminated tumor cell reservoir. CTCs can give rise to new metastatic lesions and metastases are responsible for most cancer-related deaths.
While both ctDNA and CTCs are potential biomarkers for cancer development and therapy resistance, the two are found at different stages of tumor development.
Benefits of ctDNA and CTCs for Cancer Detection
There are benefits to both ctDNA and CTCs for early cancer detection. ctDNA tends to be easier to isolate and has high sensitivity when compared to CTCs. However, CTCs have the potential to provide further insight into how cancer metastasizes.
Challenges of ctDNA and CTCs
Each presents its own challenges in detection. Both ctDNA and CTCs require large sample sizes for accurate analysis. CTCs are also heterogenous, making characterization and overall analysis challenging.
Liquid Biopsy Testing with ctDNA and CTCs
Liquid biopsy testing enables researchers to study and detect specific mutations in DNA that come from cancerous cells in plasma, urine, serum, and cerebrospinal fluid. Because liquid biopsy only requires a simple blood test and is therefore considered to be non-invasive, it is easy to collect samples from individuals. Liquid biopsy can help with the early detection of cancer, help to create customized levels of more effective treatments and therapies, and identify potential resistance mechanisms.
ctDNA places an emphasis on targeting and identifying pieces of DNA from tumor cells that are circulating in the blood.
This type of testing has the below advantages:
- Early detection of cancers and therefore early diagnosis
- Minimally invasive, only requiring a blood test (whereas tissue biopsies might require surgery)
- Creating customized treatment plans
- Identifying when treatments are effective or ineffective
- Detecting any remaining cancer or relapse
- Rapid results from NGS testing
Challenges of Liquid Biopsy
Despite having many benefits, there are several challenges associated with the use of liquid biopsy. First, it is an area that is being actively developed. More data and studies are needed to further develop an understanding of cancer biology through liquid biopsy. There is also a risk of false positives or false negatives due to low tumor DNA shed and/or tumor DNA shed from normal cells.
Second, accurate detection of rare mutations is difficult, as the extremely low fraction of cell-free DNA (cfDNA) samples is a critical challenge.
Next-Generation Sequencing For Analyzing ctDNA and CTCs
Next-Generation Sequencing (NGS) has made it possible to detect a large number of mutations in many genes on many samples. By using targeted NGS panels, sequencing can be focused on clinically relevant targets so that each target is sequenced thousands or tens of thousands of times to ensure a high degree of sensitivity. High specificity is achieved using error-correction techniques to remove errors that arise from sequencing and amplification of the DNA.
The CleanPlex® UMI Lung Cancer Panel is a targeted resequencing assay designed for rapid and high confident detection of low-frequency variants across the hotspot regions of 23 genes associated with lung cancer. Based on CleanPlex® UMI technology, this panel is specifically designed to work with cell-free DNA (cfDNA) and FFPE DNA for precise analysis of tumor DNA in liquid biopsy and tumor profiling applications.
The CleanPlex® OncoZoom® Cancer Hotspot Kit is a multiplex PCR-based targeted resequencing assay designed for rapid cancer profiling of somatic mutations across the hotspot regions of 65 oncogenes and tumor suppressor genes. Starting with just 100 pg of high-quality genomic DNA, sequencing-ready libraries can be prepared using a single-tube workflow in just 3 hours. The panel is optimized to deliver data with high on-target performance and high coverage uniformity to ensure efficient use of sequencing reads.
Product Highlight
For researchers interested in liquid biopsy, the Paragon Genomics CleanPlex UMI Technology provides highly confident detection of low-frequency variants, which can be applied to a liquid biopsy workflow. Specifically, those below 0.5% allele can be detected using just 30ng of input DNA.
View ProductFree Consultation
Learn more about our technology uses for applications such as liquid biopsy
Enter your email below to schedule a free consultation with our expert scientists to learn how our best-in-class panel can help advance your work.
Related Blog

Overview of Next-Generation Sequencing, Targeted Sequencing and CleanPlex Technology for Precision Medicine and Research

Overview of Next-Generation Sequencing, Targeted Sequencing and CleanPlex Technology for Precision Medicine and Research Select Options

